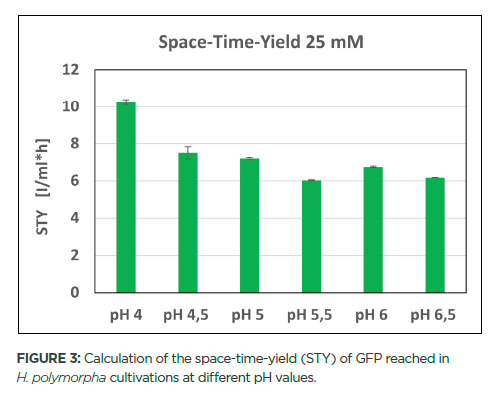
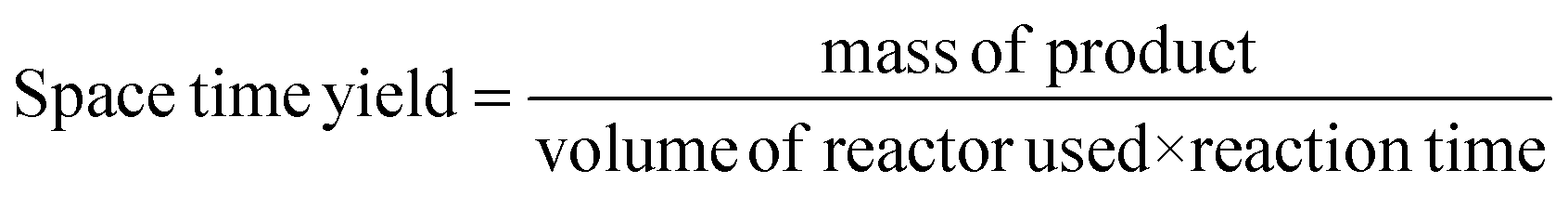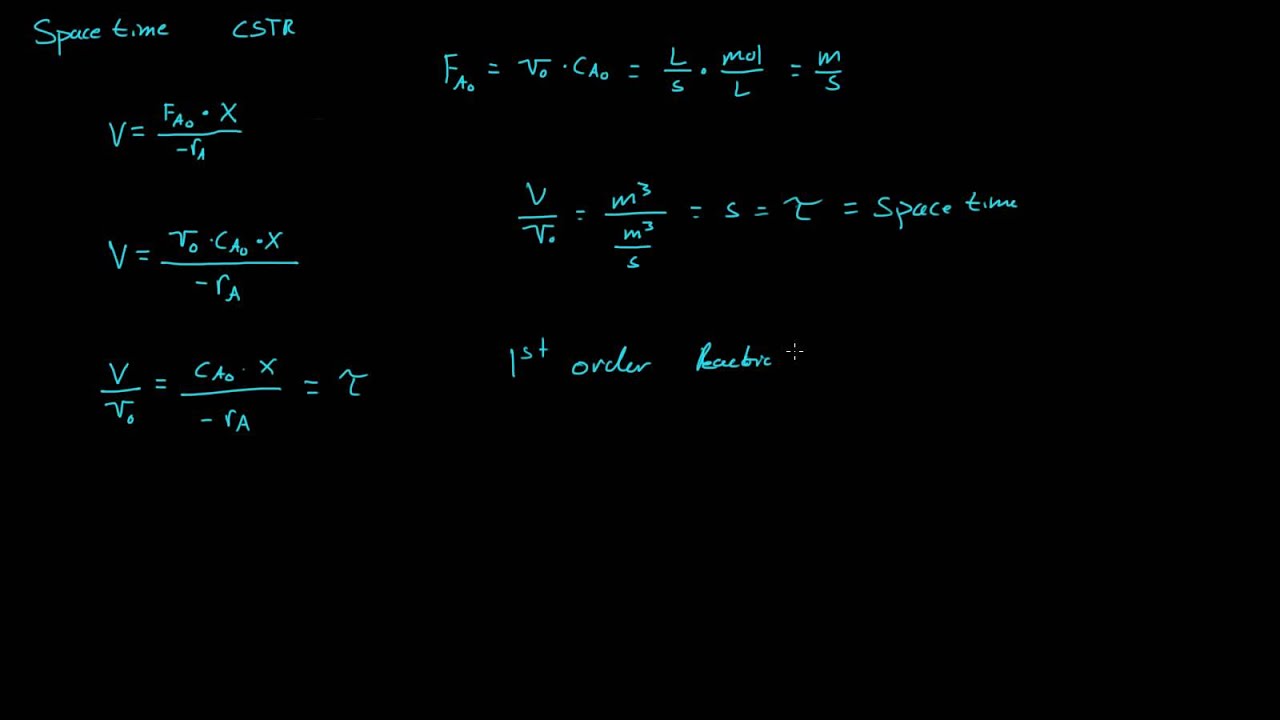A) space-time yield (STY) per kilogram of catalyst in the HCl oxidation... | Download Scientific Diagram

A) space-time yield (STY) per kilogram of catalyst in the HCl oxidation... | Download Scientific Diagram

Methods to calculate space averaged concentrations. (A) The time and... | Download Scientific Diagram

SOLVED: 3. Introduction to multiple reactions. The liquid phase reactions k1 A B are taking place in an ideal CSTR. Reaction (1) is reversible; Reaction (2) is irreversible.B is the desired product.